Keep Your Distance
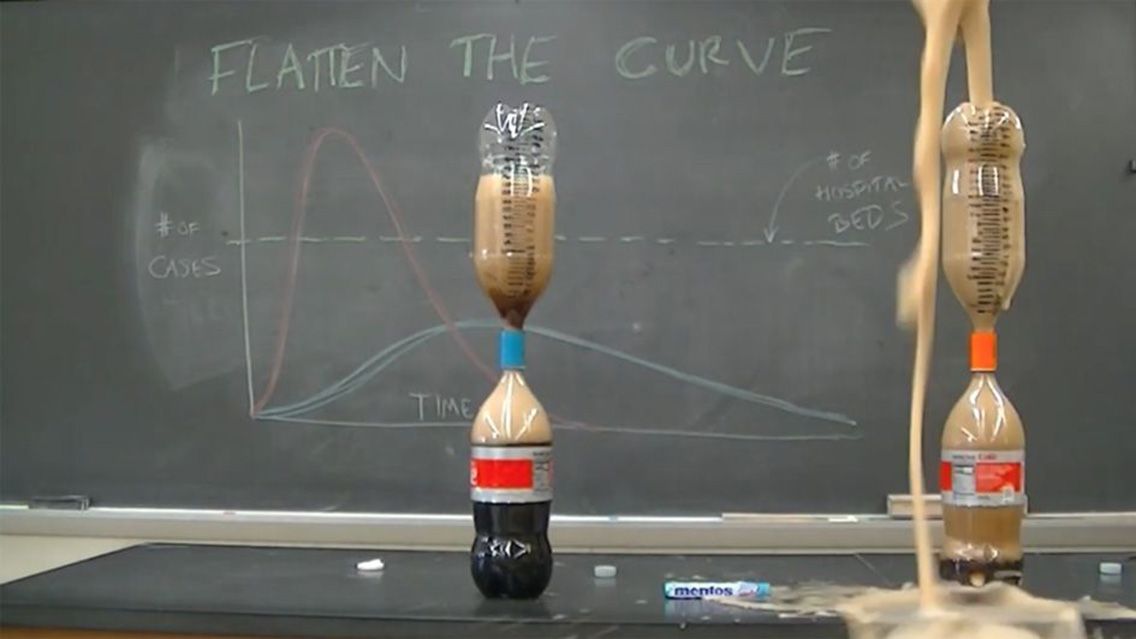
We’ve all heard slowing the spread of COVID-19 infections through social/physical distancing helps medical professionals stretch resources and deal with the pandemic. Charts and graphs are repeatedly used to demonstrate this, but is there a more visual way to bring that message home?
Sensing chemical reactions could illustrate this power, chemistry professor Dean Campbell had an idea.
“I realized that this (flattening the curve) was reminiscent of chemical kinetics: higher people concentration produces faster illness spread much like higher reactant concentrations produce faster chemical reaction,” he said.
Campbell was working with students on demonstrations involving hydrogen peroxide and iron beads, using them to illustrate graphically how adding water and diluting the peroxide (representing limits on interactions) reduces the resulting chemical reaction so it doesn’t overflow the container (the health care system).
The undiluted peroxide reacts with the same amount of iron beads and causes the container to overflow as the peroxide is converted to oxygen gas foam at a faster rate.
“My students have experience with these and other demonstrations, both by doing research with me and by doing science outreach with the Bradley Chemistry Club Demo Crew,” Campbell said.
While seniors Cassidy Kraft, Emily Rosengarten and Kayla Lippincott weren’t able to attend the experiments, which were done mostly at Campbell’s home during the extended spring break, they did remotely edit the write-up published in Chemical Education Xchange, an online resource for chemistry education.
“I'm more of a visual learner, so seeing the impact social distancing has on the health care system showed me the importance of staying home as much as possible,” Lippincott said.
“This idea was discussed right as the Bradley campus was closing for spring break and the subsequent quarantine,” Rosengarten added. “The experiments … effectively convey the fact that limiting social interactions during this pandemic can help contain the number of severe cases within the capacity of hospitals and medical equipment.”
Included in their video and the accompanying paper was a similar experiment using Mentos candy and Diet Coke done by Campbell’s colleague, Thomas Kuntzleman of Spring Arbor College in Michigan.
View videos of the experiment and the accompanying paper.
— Bob Grimson ‘81